Here’s a story that should help you remember the order of metals in the reactivity series, from top to bottom, most reactive to least reactive: It does not store any personal data.Remembering the reactivity series of metals Method 1 The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. The cookie is used to store the user consent for the cookies in the category "Performance". This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. The cookies is used to store the user consent for the cookies in the category "Necessary". The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". The cookie is used to store the user consent for the cookies in the category "Analytics". These cookies ensure basic functionalities and security features of the website, anonymously. Necessary cookies are absolutely essential for the website to function properly. There is, however, a better memory method to memorize and this makes sure you never forget. A more effective way to memorize the reactivity series is the story-link techniques. 
The simplest mnemonic for remembering the chemistry reactivity series is using the acrimony memory method: Although simple may not always be effective. What is the simplest mnemonic for remembering the reactivity series? Now the question is, why do i say so? Simple, solely due to the fact that they depend upon ligands and metals. To be honest, i would personally not recommend a mnemonic on spectrochemical series. Do you recommend a mnemonic for spectrochemical series? Remember the time you woke up whole night remembering it and the next morning the elements go upside down. A series of elements we come across time and often. I’ll provide you a simple mnemonic for remembering THE ELECTROCHEMICAL SERIES. How do you remember the electrochemical series? Mercury – Hg – Milli ( m from mercury) Silver – Ag – silver. READ ALSO: What is the point of Google stadia? What is a mnemonic for the reactivity series? 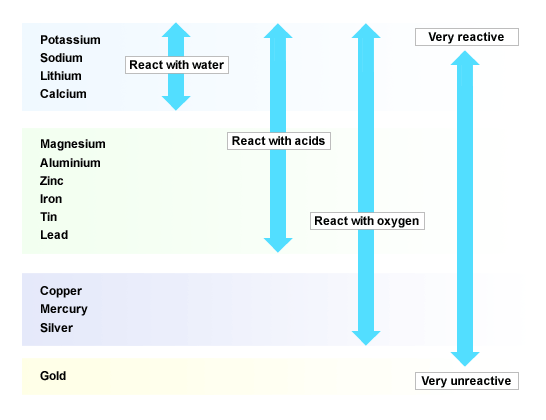
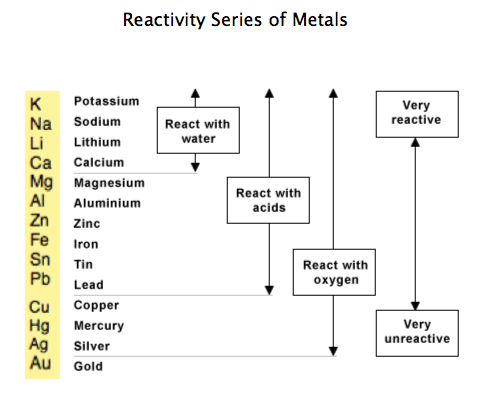
A redox reaction is feasible only if the species which has higher potential is reduced i.e., accepts the electrons and the species which has lower reduction potential is oxidized i.e., loses electrons. Is electrochemical series same as reactivity series? How does electrochemical series help in predicting the feasibility of redox reaction?Īnswer: From the E° values of the two electrodes one can find out whether a given redox reaction is feasible or not. While metals from magnesium to lead can react with acids….Short Trick to Remember Reactivity Series – Elements of Reactivity Series Metals from potassium to calcium are highly reactive and even react with water. What is the easiest way to remember the reactivity series of metals? Like calcium is more reactive than magnesium. Reactivity series is about the reactivity of the chemical substances in a particular reaction. On the other hand, reactivity series is for the reaction without being in aqueous state. READ ALSO: Can you take chocolate into NZ? Is electrochemical series and reactivity series Same?īasically, ECS (electrochemical series) is to refer for aqueous state.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
